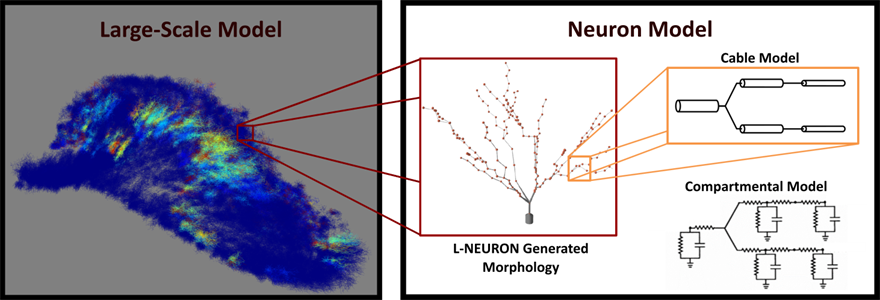
The main goal of the large-scale modeling project is to investigate how interactions of all neural properties affect the input transformation properties of the hippocampus and how these transformations enable it to play its crucial role in memory formation. In order to construct the model, we consider several properties that are common to neuron populations but specify the parameters uniquely to the rat hippocampal system. These properties include large numbers of neurons, the unique morphologies that differentiate neuron types, the distributions of membrane channels that determine the response characteristics of neurons, the topographical connectivity between neurons, and the activity-dependent non-stationarities that modulate the strengths of synapses over time. Using the NEURON simulation environment, multi-compartment models of neurons are constructed and embedded with membrane channels that imbue the model with realistic electrophysiological characteristics. Different synaptic receptors can be inserted to mediate communication between neurons, and various plasticity rules and mechanisms can be implemented to modulate the synaptic weights. L-NEURON is used to generate the dendritic structure characteristic to the different cell types of the hippocampus. With approximately 200 compartments per neuron and over 1.5 million neurons per hippocampal formation, the simulations are parallelized and run using a dedicated high performance computing cluster. The large-scale modeling framework allows us to incorporate data from many sources and, in effect, transforms the model into a functional compendium that can represent as much detail as is reasonable from the vast hippocampal literature. The flexibility and detail of the model allows us not only to investigate how the interactions of these properties may cause different functions to emerge but also to test current hypotheses in hippocampal function such as pattern separation, pattern completion, and place field formation and, furthermore, to discover new hypotheses as well.
Selected peer-reviewed publications
2. Bingham, C.S., Loizos, K., Yu, G.J., Gilbert, A., Bouteiller, J.-M.C., Song, D., Lazzi, G., and Berger, T.W. Model-based analysis of electrode placement and pulse amplitude for hippocampal stimulation. IEEE Transactions on Biomedical Engineering, 2018, 99, 1-1, DOI: 10.1109/TBME.2018.2791860.
3. Hendrickson, P.J., Yu, G.J., Song, D., and Berger, T. W. Interactions between inhibitory interneurons and excitatory associational circuitry in determining spatio-temporal dynamics of hippocampal dentate granule cells: A large-scale computational study. Frontiers in Systems Neuroscience, 2015, DOI: 10.3389/fnsys.2015.00155.
4. Hendrickson, P.J., Yu, G.J., Song, D., and Berger, T. W. A million-plus neuron model of the hippocampal dentate gyrus: Critical role for topography in determining spatiotemporal network dynamics. IEEE Transactions on Biomedical Engineering, 2015, 63(1), DOI: 10.1109/TBME.2015.2445771.
5. Yu, G.J., Hendrickson, P.J., Song, D., and Berger, T.W. Spatio-temporal patterns of granule cell activity revealed by a large-scale, biologically realistic model of the hippocampal dentate gyrus. In: Hippocampal Microcircuits, Vol. II (Eds. Cutsuridis V., Graham B.P., Cobb S., and Vida I.), New York: Springer, in press